Packaging Evaluation And E&L Testing For Optimal Drug Quality
What Are Extractables And Leachables?
Mutual interactions between the pharmaceutical and packaging are a problem with pharmaceutical packaging materials that require particular care. Extractables are compounds generated from storage conditions that are more severe than normal, whereas leachables are compounds that can be transferred from the packaging to the pharmaceutical under normal storage conditions. They are classified as indicated in Table 1. Before pharmaceuticals can be sold, extractables and leachables must be comprehensively verified to determine packaging material risks.
| Extractables | Leachables | |
|---|---|---|
| Description | All compounds that potentially could be extracted from packaging | Compounds that leach out under normal usage conditions |
| Extraction Conditions | More severe conditions than for normal use or storage. High-temperature extraction or solvent extraction is used. | Under normal conditions for use or storage. |
| Measured Item | Packaging material | Pharmaceutical |
The Importance Of Extractables And Leachables Studies

Drug product efficacy, safety and quality are largely dependent on their integrity and purity. Extractables and leachables studies strive to minimize the possibilities of contamination that taint or damage the composition of drug formulations, thereby ensuring patient safety. If not monitored or controlled, extractables and leachables can pose severe health risks, such as toxicity and carcinogenicity.
It is important to note that extractables and leachables studies should not be isolated to the final drug product alone. Medical devices, drug delivery devices and container closure systems should all be evaluated for the presence of extractables and leachables. The migration of unwanted chemicals can happen anytime during the drug product manufacturing process, even after the packaging system following drug formulation.
What Are The Standards Involved In Testing E&L?

There are various standards for extractables and leachables, and the United States Pharmacopeia (USP), PQRI, BPOG, etc. have published their own documents, but there is no unified guideline. To overcome this situation, ICH established a new working group for the evaluation and management of extractables and leachables in July 2020 and reached Step 1, or ICH-Q3E, in 2020, and aim to reach Step 4 in 2024.
This is in particular to the rise of biopharmaceuticals, where its increase in use has drawn attention to the following:
- The transfer of chemical substances by storage
- The transfer of chemical substances by manufacturing
In its “Guidance for Industry Container Closure Systems for Packaging Human Drugs and Biologics”, the FDA refers to the potential interactions between drugs and packaging materials. They claim that injectable and injectable suspension formulations pose a high risk to the human body from E&L (Table 3). As of 2021, biopharmaceuticals accounted for more than 30% of the total pharmaceutical market, and evaluation of E&L has become increasingly important. Biopharmaceuticals are difficult to take orally because of their high molecular weight, so most of it is injectable. In other words, biopharmaceuticals pose more concerns when it comes to E&L than small molecule drugs.
In addition, the cost of R&D and manufacturing of biopharmaceuticals is high, therefore, to decrease development costs and production facilities there is a growing momentum for continuous production of multiple products on a small scale, as opposed to the conventional large-scale production facility using large stainless steel culture tanks. However, since these types of production facilities use single-use equipment made of resin material, risk evaluations of leachables generated between the resin used in the facility and the pharmaceuticals must be implemented.
| Degree Of Concern Associated With The Route Of Administration | Likelihood Of Packaging Component Interaction Dosage | ||
|---|---|---|---|
| High | Medium | Low | |
| Highest | Inhalation aerosol and Solutions; Injections and Injectable Suspensions |
Sterile Powder and Powders for Injection; Inhalation powder |
|
| High | Ophthalmic Solutions and Suspensions; Transdermal Ointments and Patches; Nasal Aerosols and Sprays |
||
| Low | Topical Solutions and Suspensions; Topical and Lingual Aerosols; Oral Solutions and suspensions |
Topical Powders; Oral powders |
Oral Tablets and Oral (Hard and Soft Gelatin) Capsules |
Is There a Unified Method To Test for Extractables and Leachables?
Unfortunately, having a single method that covers all chemical substances in the analysis is extremely difficult. Chemical substances (monomers, polymerization initiators, antistatic agents, adhesives, etc.) contained in packaging materials and single-use products are often not disclosed due to the unique technologies, knowledge, and patents of each manufacturer. Currently, ICH Q3E is under consideration and aiming to set a threshold value similar to ICH-Q3A / B. Discussions are underway on thresholds that need to be reported, as well as thresholds that require structural determination, and also thresholds that require safety confirmation.
Analytical Techniques In Extractables And Leachables Testing

USP1663 recommends cooperation with vendors (suppliers). This is because the chemicals contained in packaging materials and single-use products are generally unclear. In order to efficiently discover, identify, and quantify leachables, identify the main sources together with the vendor, and determine your experimental parameters, taking in account extraction variables (temperature, time, area to volume ratio, solvent, etc). It may be recommended that users can choose the measurement method that best suits the substance by adding the predicted substance as an extractable/leachable. Shimadzu provides a wide range of equipment and solutions needed to measure E&L. For example, In the process of measuring volatile and semi-volatile organic compounds, the optimal solution is GC/MS. Solvent extraction and heat extraction methods can be used followed by GC/MS for analysis. In contrast, for measuring refractory organic compounds, LC and LC/MS provide an alternative solution that enables the identification and quantification of non-volatile organic matter. Finally, to measure inorganic extracts, ICP, ICP/MS, AA, and other similar methods are suitable. These methods are effective for measuring inorganic (element) extracts.
Explore Our Solutions
Physical Integrity Of Pharma Packaging
Evaluating Physical Strength: Product And Packaging Testing
While chemical safety is critical, the physical integrity of both the drug product and its packaging is equally important. Rigorous physical testing ensures that medications can withstand the stresses of manufacturing and transport, and that the packaging protects the product while remaining functional for the end-user. This dual focus on product and packaging integrity is fundamental to delivering a safe and effective pharmaceutical product.
Key Testing Methods For Product Integrity
These tests focus on the physical characteristics of the drug formulation itself to ensure it meets quality standards for durability and performance.
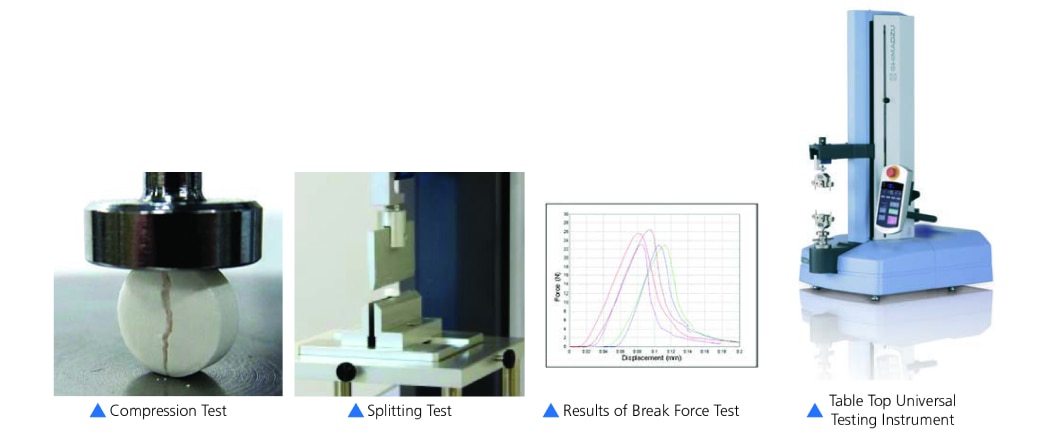
Pill Compression Test
The Pill Compression Test evaluates the hardness and structural integrity of tablets. A tablet must be robust enough to avoid cracking or splitting during production, packaging, and shipping. However, if it is too hard, it may not dissolve correctly in the body, which could reduce its effectiveness. This test measures the force required to crush a tablet, providing essential data for quality control. For tablets designed to be split, the test also confirms that they break cleanly along the scored line with a suitable amount of force, ensuring accurate dosing.
How It Is Performed:
- Pill crush test is performed in order to understand the compressive properties of tablets. Evaluate in terms of hardness, powder molding, and surface coating characteristics.
- Splitting test is performed on tablets with grooves in the center, allowing each to be taken whole by adults or in a half dose by children.
- For this reason, it must be split in half using some suitable degree of force.
Pellet Crush Test
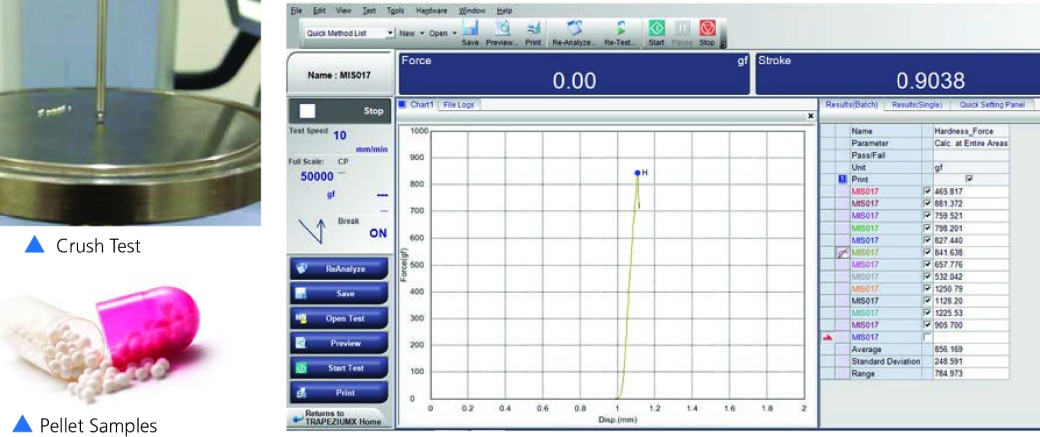
Similar to the pill compression test, the Pellet Crush Test is designed to measure the crushing strength of individual pellets, which are often used in controlled-release drug formulations. The primary objective is to determine the force at which the pellet's outer coating fails. This is crucial for quality control, as the coating's integrity is directly linked to the drug's release profile and overall efficacy. The test helps ensure that the pellets remain intact until they reach the intended point of action in the body.
How It Is Performed:
- The objective of testing is to measure the crushing strength of pellets at which failure occurs.
- The peek force i.e., Force required to crush the pellet coating is marked on the graph and readings are recorded in the software. Refer here.
- Our testing and observation: When 10 gm pre-load force is detected, probe further crush the sample to measure the maximum peak and stop the test once the load drops to the 80% of maximum hardness value.
Key Testing Methods For Packaging Integrity
This test focuses on the functionality and usability of the final drug package.
Press-Dispense Test
The Press-Dispense Test is a quality control measure for press-through packages (PTPs), commonly known as blister packs. It evaluates the force required for a patient to push a capsule or tablet through the foil or plastic backing. The goal is to find the right balance: the seal must be strong enough to protect the product, but not so strong that it becomes difficult for a patient, especially those with limited strength, to dispense the medication. This test ensures the packaging is both secure and user-friendly, which is a critical aspect of patient adherence and overall product quality.
How It Is Performed:
- The objective of testing is to measure the crushing strength of pellets at which failure occurs.
- The peek force i.e., Force required to crush the pellet coating is marked on the graph and readings are recorded in the software. Refer here.
- Our testing and observation: When 10 gm pre-load force is detected, probe further crush the sample to measure the maximum peak and stop the test once the load drops to the 80% of maximum hardness value.



